|
mass of a neutral boron atom 10.012 939 u mass of a neutral lithium-7 atom 7. In fact, it decays by both spontaneous fission and alpha emission, in a 97:3 ratio. Boron undergoes fission via thermal neutron capture to produce lithium-7, an alpha particle and energy in the following reaction: nB LiHe+ 5 A + 10 3 7 2 4 Using the data below, calculate mass defect (in u and kg) and binding energy (in J and MeV) for this reaction. These fragments, or fission products, are about equal to half the original mass. The fission of 1 kilogram of uranium-235, for example, produces about 2.5 million times as much energy as is produced by burning 1 kilogram of coal.\) will decay by either or both of these two processes. When a nucleus fissions, it splits into several smaller fragments. Examples that must be solved start at 11. The approximate time elapse between stages of the process is indicated at the bottom of the Figure. 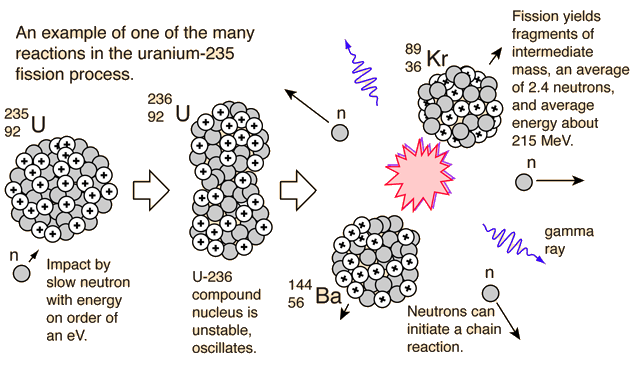
Nuclear fission reactions produce incredibly large amounts of energy compared to chemical reactions. The first ten examples are different fission reactions with some comments attached here and there. Nuclear fission - Stages, Reactions, Energy: A pictorial representation of the sequence of events in the fission of a heavy nucleus is given in Figure 3. Chain reactions are one way that systems which are not in thermodynamic equilibrium can release energy or increase entropy in order to. This is an extremely important aspect of fission, because neutrons can induce more fission, enabling self-sustaining chain reactions. 
Most fission produces neutrons, although the number varies. In a chain reaction, positive feedback leads to a self-amplifying chain of events. Fission is a statistical process in which an entire range of products are produced with various probabilities. In uncontrolled nuclear fission, one fission reaction starts a chain reaction, in which neutrons produced in one reaction cause other reactions, which cause more reactions, and so on. This energy is a result of the conversion of a small amount of mass into energy, as described by Einstein’s famous equation, Emc². It begins when the nucleus of a radioactive atom gains a neutron. 
For instance, when one mole of U-235 undergoes fission, the products weigh about 0.2 grams less than the reactants this “lost” mass is converted into a very large amount of energy, about 1.8 × 10 10 kJ per mole of U-235. A chain reaction is a sequence of reactions where a reactive product or by-product causes additional reactions to take place. This type of reaction releases a great deal of energy from a very small amount of matter. (b) The larger fission products of U-235 are typically one isotope with a mass number around 85–105, and another isotope with a mass number that is about 50% larger, that is, about 130–150.Ī tremendous amount of energy is produced by the fission of heavy elements. He derived an expression for the stationary fission rate (or reaction rate in chemistry) as a function of the temperature, the friction. \): (a) Nuclear fission of U-235 produces a range of fission products.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
